The Fund for LSSU
Automate 2019
LSSU Students
School of Engineering & Technology
The LSSU Engineering students that attended Automate 2019 were selected competitively based on academic success and approval from LSSU engineering professors. These students are listed in Table 1 and are shown representing LSSU at our Automate 2019 conference booth in Fig. 1.
Table 1 – LSSU students selected to attend Automate 2019.
| Name | Class Standing | Major |
| Brandon Niemi | Senior | Electrical Engineering |
| Alex Alias | Senior | Manufacturing Engineering Tech |
| Rick Miller | Senior | Mechanical Engineering |
| Preston Fairchild | Junior | Electrical Engineering |
| Thomas Muir | Freshman | Mechanical Engineering |
| Josh Schember | Freshman | Computer Engineering |
| Trevor Simons | Senior | Mechanical Engineering |
| Evan Reeves | Senior | Mechanical Engineering |
| Eden LaTulip | Junior | Computer Engineering |
| Nathan Pim | Senior | Manufacturing Engineering Tech |
Fig. 1 – LSSU Students and professors at Automate 2019 pictured in front of LSSU’s conference booth. From left to right: Thomas Muir, Rick Miller, Preston Fairchild, Evan Reeves, Brandon Niemi, Alex Elias, Trevor Simons, Nathan Pim, Eden LaTulip, Prof. Jim Devaprasad, and Prof. Edoardo Sarda
Fig. 2 – Robotics Professionals Watching Demonstrations at LSSU’s Booth
The Automate 2019 conference was held at McCormick Place in downtown Chicago, IL from Monday, April 8, 2019 to Thursday, April 11, 2019. Our group attended events on all the days of the conference. The event was divided into two parts: the trade show, captured in the panoramic photo shown if Fig. 3 below and the conference sessions, which were hosted on the floor beneath the show.
Fig. 3 – Panoramic view of the Automate trade show floor
Attendance of the trade show exhibition was free, however the conference sessions required passes which were purchased using the money provided by the Fund for LSSU grant. Each student was asked to pick two to three conference sessions that he/she wanted to attend based on interest, and the conference passes were purchased accordingly (see Table 2).
Table 2 – Conference Pass Distribution per Attendee.
| Name | Conference Dates Attended |
| Brandon Niemi | April 8th & 11th |
| Preston Fairchild | April 8th & 11th |
| Alex Elias | April 8th & 9th |
| Rick Miller | April 8th & 9th |
| Thomas Muir | April 8th & 9th |
| Josh Schember | April 8th & 9th |
| Trevor Simons | April 10th & 11th |
| Evan Reeves | April 10th & 11th |
| Eden LaTulip | April 10th & 11th |
| Nathan Pim | April 10th & 11th |
A sample of the sessions that were attended throughout the four days along with a brief description of what each conference session discussed is detailed below:
- Monday
- The Growing Importance of Software in Manufacturing
Maxwell Reynolds, Co-founder, CEO Symbio
While Artificial Intelligence, machine learning, and modern networking infrastructure are setting the stage for a revolution in industrial manufacturing, engineers today cannot take full advantage of these technologies because robotics and sensor platforms are dependent on vendor specific proprietary software. Our presentation will provide basic definitions and context to advanced technologies and show how their application on the factory floor will increase robot capabilities and enable flexible workcells.
- Tuesday
- Choosing the Right Vision Solution
Instructor: David Dechow, Principal Vision Systems Architect, Integro Technologies
One of the fundamental challenges in implementing machine vision technologies in automation applications is the selection of the right solution. Whether the task is to identify and apply the machine vision components, or to specify and purchase an entire machine vision-based automation cell, there are some basic steps and processes to follow that can help ensure the success of your project. In this presentation we will discuss machine vision application analysis and system specification related to key machine vision technologies. The presentation will also outline typical machine vision components and software along with the applications they enable and the potential limitations to be aware of. The presentation is valuable for all attendees who use, specify or plan to implement machine vision in their automation environment.
- Automation: Off-the-Shelf vs. Custom?
Presenter: Paul Santi, General Manager – Automation Systems Group, FANUC America
Standard, off-the-shelf automation may work for some applications. However, as Abraham Maslow said, “If you only have a hammer, everything looks like a nail.” We have all witnessed that a hammer does not always work — even for every type of nail. Right Sizing the automation solution and selling the right size is critical to achieving long-term value results. This session will outline methods to Right Size and Value Justify custom automation solutions with and without Collaborative Technologies.
- Wednesday
- Higher & Faster — New Vision Architectures to Solve Complex Challenges
Presenter: Thomas Karow, Product Market Manager, Basler AG
There is an ongoing trend for higher resolution and faster framerates to steadily increase performance in vision systems. Let’s divide the challenges into the parts of the sensor, camera interface and data processing. The timing is good to think about. Now! Technologies have ripened to cover those needs and give answers to these challenges. The presentation covered all these points, make it clear to the audience how the needs lead to challenges. Moreover, how they can be addressed with solutions.
- Intro to Automation & Robotics on the Factory Floor
Presenter: Jason Claggett: Business Solutions, Encompass Solutions
The presentation serves as an introduction to manufacturers who have little-to-intermediate knowledge of how automation and robotics are used in manufacturing settings. This included a brief overview of the history of automation, how we’ve arrived at the current state of manufacturing (industry 4.0) and what types of technology we are seeing emerge.
- Thursday
- Collaborative Robot Safety Overview & Risk Assessment
Panel included:
Carole Franklin: Director of Standards Development, Robotics Association for Advancing Automation
Elena Dominguez: Safety Consultant Pilz Automation Safety
Collaborative robot systems are a new robotics technology that allows for robots and humans to work together in ways that were previously impossible. However, with this new capability comes new hazards and risks that must be evaluated for every application. This session introduced key concepts in collaborative robot system safety, including introduction to relevant safety standards, and how to evaluate and mitigate these new hazards related to collaborative robot applications.
Ms. Franklin opened the session with an introduction to robot safety standards and concepts, and how collaborative robotics has changed the safety paradigm.
Ms. Dominguez discussed the collaborative robot Risk Assessment process in detail and gave a worked example.
The conference sessions provided a refreshing real-world angle on concepts that the students had studied in their coursework at LSSU. In addition to providing an enlightening supplement to coursework, the conference sessions gave the students a chance to meet and interact with industry professionals. It was opportunities like the events at Automate 2019 that made attending the conference sessions worthwhile for more than just professional training.
When not attending conference sessions, members of the group took turns representing LSSU at LSSU’s conference booth and exploring the trade show floor, which was full of engaging demonstrations of the newest technology in the industry.
Even though LSSU’s robotics program is notable, the students and faculty would not have been exposed to this type of emerging technology without attending Automate 2019. Furthermore, LSSU was the only university that had a booth in the conference. This drew special attention from many of the industry professionals that allowed interaction between LSSU students and faculty resulting in great conversations that showed well on behalf of both the students and LSSU altogether.
Overall, Automate 2019 was an unforgettable experience that allowed LSSU students and faculty to explore the latest trends and technology in the industry.
In close, the students and faculty from the School of Engineering and Technology are very appreciative of getting the opportunity to represent LSSU at the Automate 2019 conference and are very thankful to the “Fund for LSSU” for supporting this trip by funding the travel to and from Automate along with the conference registrations.
Campus Veterans Fair
LSSU Students
Nursing 432
 The Veteran Resource Open House was held on March 20, 2019 in the Superior Room of
The Veteran Resource Open House was held on March 20, 2019 in the Superior Room of
the Cisler Center. Upon entering the event, the veterans were presented with a name tag that was
color coded for the branch of the military in which they served but had their name and
country/area(s) where they had served written on them. The name tags were purchased with
grant funding. Several exhibitors were invited to set up booths at the event such as admissions,
Community Action, Veterans Affairs, etc. Our class put together a few poster boards on tips to
communicate with your physician and the differing health risks and exposures that resulted from
various battle sites. The poster supplies were purchased by grant funding. Our class also
provided a booth of health screenings; blood pressure and blood glucose. The grant funding
purchased gloves and cotton balls for blood glucose supplies while the school of nursing
provided glucometers, test strips, alcohol pads, etc. The class had also gathered donations from
businesses around the Soo and we purchased raffle tickets and paper bags to create drawings for
the prizes. The grant funding also gave the NURS 432 students the opportunity to purchase two
$75 Meijer gift cards and one $100 VISA card to raffle off as grand prize awards. A cake was
purchased with grant funding that had “Thank you for your service” in script on it that was
enjoyed by veterans and the NURS 432 students.
Thank you again for awarding the NURS 432 class with the Enrichment Grant as this had
provided us with funding for our Community Health project. The products and prizes that were
purchased using the funding from this grant contributed to the events success and helped entice
veterans to come gain education regarding their health and learn about resources on campus and
in the community that are available to them.
Carolyn Turgeon Visiting Writer project experience
Mary McMyne
Associate Professor, School of Arts and Letters
 The LSSU Creative Writing Program is grateful to the LSSU Foundation for the $1,000 speaker fee/honorarium which was mailed to Carolyn immediately upon her visit. Her visit was a resounding success with a number of students and community members attending her various talks and lectures, and a crowd at the library for her reading. Books were sold and copies were signed as well. Thank you for your support of the LSSU Creative Writing Program and its students
The LSSU Creative Writing Program is grateful to the LSSU Foundation for the $1,000 speaker fee/honorarium which was mailed to Carolyn immediately upon her visit. Her visit was a resounding success with a number of students and community members attending her various talks and lectures, and a crowd at the library for her reading. Books were sold and copies were signed as well. Thank you for your support of the LSSU Creative Writing Program and its students
GSA North-Central Sectional Meeting
 The purpose of the travel was to present credible research at a national sectional meeting in Manhattan, KS. The research was to show past river migration patterns from aerial photographs of the Lower Muskegon River in Michigan. Attendance at this sectional meeting allowed for the data from this research project to become made public to the rest of the nation to understand human impacts on river systems in Michigan. Attendance was also crucial because it allowed for increasing professionalism in the geological sciences. In other words, we met with professional geologists and created networks between different groups of people to expend our connections with other geologists. The follow pictures were taken at the meeting (photo credit: MaryKathryn Rocheford):
The purpose of the travel was to present credible research at a national sectional meeting in Manhattan, KS. The research was to show past river migration patterns from aerial photographs of the Lower Muskegon River in Michigan. Attendance at this sectional meeting allowed for the data from this research project to become made public to the rest of the nation to understand human impacts on river systems in Michigan. Attendance was also crucial because it allowed for increasing professionalism in the geological sciences. In other words, we met with professional geologists and created networks between different groups of people to expend our connections with other geologists. The follow pictures were taken at the meeting (photo credit: MaryKathryn Rocheford):
Tri-regional Geological Society of America(GSA) meeting
Matthew Bell
Student, College of Science and the Environment
 With the help of the LSSU Foundation, I attended the Tri-regional Geological Society of America(GSA) meeting held in Manhattan, KS from March 24th-26th. My objective of attending was to present the results of my research to the scientific community and make a valid contribution to the technical program; and learn from (and network with) professionals who are experts in their fields. I was able to represent LSSU on a nearly national level and gain exposure to the geopolitical aspect of my field. This was an extremely beneficial experience (personally, professionally, and academically) and I am grateful to have lived it.
With the help of the LSSU Foundation, I attended the Tri-regional Geological Society of America(GSA) meeting held in Manhattan, KS from March 24th-26th. My objective of attending was to present the results of my research to the scientific community and make a valid contribution to the technical program; and learn from (and network with) professionals who are experts in their fields. I was able to represent LSSU on a nearly national level and gain exposure to the geopolitical aspect of my field. This was an extremely beneficial experience (personally, professionally, and academically) and I am grateful to have lived it.
Development of a Biologically Based Screening Method for Perfluoroalkyl Substrates and Other Contaminants of Emerging Concern.
Britton Ranson Olson
Associate Professor, School of Biological Sciences
PROJECT SUMMARY
We have developed a model to identify the cellular and molecular toxicity targets of perfluoroalkyl  substrates (PFASs). These are manmade, biologically recalcitrant chemicals which are considered ubiquitous contaminants of the environment and have been implicated with adverse health effects. We are working to develop this model into one that serves as a general cellular model, having the universal components and features of all cells (a semi-permeable membrane enclosing biosynthetic enzymes and other proteins, ribosomes, and so on), but also, because it possesses both of the central energy metabolisms that are distributed among animals and plants, a broad, biologically based screening method for detecting contaminants of emerging concern.
substrates (PFASs). These are manmade, biologically recalcitrant chemicals which are considered ubiquitous contaminants of the environment and have been implicated with adverse health effects. We are working to develop this model into one that serves as a general cellular model, having the universal components and features of all cells (a semi-permeable membrane enclosing biosynthetic enzymes and other proteins, ribosomes, and so on), but also, because it possesses both of the central energy metabolisms that are distributed among animals and plants, a broad, biologically based screening method for detecting contaminants of emerging concern.
The objective of this funded proposal was to advance our model as a diagnostic tool for assessing the impact of contaminants on mitochondrial function. We did so by assaying for energy production (ATP) in the presence and absence of PFASs at concentrations typical of those found within the environment and compare those measures to those of the eukaryotic model, Saccharomyces cerevisiae (baker’s yeast, possesses mitochondria). Fulfilling the pillar of “Student Professional Development”, LSSU students have and will continue to benefit from this project through the opportunity for senior thesis projects and elevated laboratory experiences as described herein.
The kits provided from the Fund were first incorporated into the laboratory component of the BIOL421 Advanced Cellular and Molecular Biology course. It is challenging to portray to students the significance of subtle data changes which are typical at a molecular level, as they are often on the micromolar, or even smaller, scale. The students were able to optimize the concentrations of mitochondria used in the assay and found that their ATP measurements reinforced existing procedures and allowed for statistical analyses that were not previously possible. The students also developed preliminary toxicology tests for various chemicals of interest. The data generated from these BIOL421 students was and will continue to be used as guide for future application to the ATP quantification studies.
The Effects of Human Activity on Sea Turtle Nesting, Incubating Egg Clutches and Hatchlings: Education through the Ethos of Conservation and Ecotourism
Madison Marsh
In association with Tortugranja Turtle Sanctuary in Isla Mujeres, Mexico while in
attendance of the Isla Mujeres Ethnographic Field School.

Taught children, locals and tourists on sustainable methods of conservation. Created ‘interactive learning’ procedures for tourists and visitors of Tortugranja. Bought 10 heads of lettuce, and free dove in the ocean pen during ‘busy time’ of day, burying lettuce in the sand. This was interactive with visitors by showing how turtles, (in their natural habitat) love lettuce as a snack!
Organized a fundraiser to be had at local restaurant, aMar Cocina Peruana for the creation and professional printing of new posters (in English and Spanish) for Tortugranja. Went business to business asking for gift
certificates or monetary donations to support the new posters and organized a silent raffle.
GLATA Meeting
SATA
Student Athletic Training Association
 The money you granted covered our registration costs for the conference, which helped us out a lot! The week was filled with many lecture based and hands on, evidence based practice educational opportunities that we would not have been able to attend without your support. While we were at GLATA, we competed in the Quiz Bowl and represented the state of Michigan. During the first half, we were leading the pack in first place, but unfortunately by the end of the competition, we finished in 4th place. This is still a great accomplishment for our school and we are very proud of our accomplishment.
The money you granted covered our registration costs for the conference, which helped us out a lot! The week was filled with many lecture based and hands on, evidence based practice educational opportunities that we would not have been able to attend without your support. While we were at GLATA, we competed in the Quiz Bowl and represented the state of Michigan. During the first half, we were leading the pack in first place, but unfortunately by the end of the competition, we finished in 4th place. This is still a great accomplishment for our school and we are very proud of our accomplishment.
On the way down, we had the opportunity to visit the Grand Rapids Griffins Stadium. We had the opportunity to tour the facility with their head Athletic Trainer, John Bernal, who is a past employee of LSSU’s Athletic Department. He provided us with knowledge about what it takes to work at a professional level and shared his experiences with us to help us get a better look at what we should expect as future professionals.
SHHHHH We have a plan
LSSU Arts Center
Over 1000 young students from the area joined us for an amazing show in the Arts Center! It was a blast. Thank you to everyone in attendance and to all those who made the day possible – LSSU Charter Schools, Fund for LSSU, and Arts Midwest Touring Fund. – For show – SHHHHH We have a plan.
USITT Conference
Jamie Nathan
Technical Director, LSSU Arts Center

This year I went to USITT in St. Louis, MO to research lighting and sound equipment that would improve the health of our technical theatre system and allow us to have higher quality tours come to Sault Ste. Marie.
First, the soundboard. I was lucky enough to get a spot in a beginning sound technician workshop where we worked on actual soundboards. The board we were using was the one that Scott and I have been looking at for the theatre, so it was a great test drive. It is a digital board with 32 channels. It is smaller and lighter than the analog board we currently utilize. It is intuitive and easy for beginners to learn. There are about three different ways to do the same function, so depending on your learning style, this board probably has a function that will work well with your brain. It is the same brand as the smaller mixer we use for movies.
I also spoke with ETC lighting. They referred me to Lighthouse in Green Bay, which would be our ‘local’ lighting dealer. They would like to do an assessment of our space and create a plan for moving us toward ETC lighting. This would help with tours that want to bring their ION boards with them and plug into our system. It would also help with some lighting challenges we’ve been having. The last big piece of information I have is that I spoke with OSHA. They have a program where they send someone out to evaluate our theatre and tell us all the safety regulations we need in place. This is done without the threat of being fined. As I understand, we would need to lop Wendy Beach into this process.
The rest of my time at the conference was spent in sessions discussing challenges of technical direction, learning more about sound mixing, and lighting for dance. I also made some contacts with other women TDs at small programs who I can troubleshoot our challenges with, and see how they grew their programs.
All of which will benefit the Arts Center at Lake Superior State University. We are moving toward a healthier technical system, I have learned some new skills that will be of great use with upcoming events, and I have started creating a support system that will help me create a quality technical theatre program for our students.
Automate 2017
LSSU Students
School of Engineering & Technology

A grant awarded by the “Fund for LSSU” allowed LSSU engineering students to attend Automate 2017, North America’s largest trade show for industrial robotics and automation technology.
The grant gave the opportunity for the students to attend a variety of conference sessions focused on everything from technical training sessions to discussions about the future of the robotics and automation industry. By attending these sessions in combination with exploring the showroom floor, the students gained priceless experience and personal connections that they can carry forward into their educational and professional careers. The LSSU Engineering students that attended Automate 2017 were selected competitively based on academic success and approval from LSSU engineering professors.
The conference sessions provided a refreshing real-world angle on concepts that we had studied in our coursework at LSSU. In addition to providing an enlightening supplement to coursework, the conference sessions gave us a chance to meet and interact with industry professionals. It was opportunities like the events at Automate 2017 that made attending the conference sessions worthwhile for more than just professional training.
When not attending conference sessions, members of our group took turns representing LSSU at our conference booth and exploring the trade show floor, which was full of engaging demonstrations of the newest technology in the industry.
Even though LSSU’s robotics program is notable, we would not have been exposed to this type of emerging technology without attending Automate 2017. Furthermore, we were only one of few universities that had a booth in the conference. This drew special attention from many of the individuals we interacted with and usually led to some great conversations that showed well on behalf of both us and LSSU altogether.
Overall, Automate 2017 was an unforgettable experience that allowed us to explore the latest trends and technology in the industry.
In close, we are very appreciative of getting the opportunity to represent LSSU at the Automate 2017 conference and are very thankful to the “Fund for LSSU” for supporting this trip by funding our lodging and conference registrations.
GLATA Meeting
SATA
Student Athletic Training Association
The faculty and students of the Student Athletic Training Association (SATA) would like to thank the LSSU Foundation for the Fund for LSSU Grant supporting registration costs for the 2017 Great Lakes Athletic Trainers’ Association (GLATA) District 4 Annual Meeting. This year’s conference was held in Wheeling, IL, March 9th – 11th, offering students and certified athletic trainers the opportunity to learn from successful athletic trainers and other health care professionals from across six states making up District 4. The conference was three days long, consisting of multiple one hour education sessions for certified and student athletic trainers. Some of our presentations involved discussion and hands-on activities to explore manual therapy techniques of the thoracic spine, nutritional guidelines for traveling athletes, neuroplasticity within athletic training, the future of orthopedic surgery, and much more. We also attended the annual GLATA quiz bowl and one of our students participated in the palpation challenge.
Having the opportunity to network with other athletic training professionals and students is vital to student development, something LSSU and the Foundation take very seriously. Austin Hawks, a recent applicant in the athletic training program at LSSU, states, “This conference was an eye-opening experience into the profession of athletic training. The networking opportunities within the great lakes region provided knowledge on current practices and techniques that I look forward to learning more about in my courses at LSSU.”
SATA would like to say thank you for supporting our education as we develop our base of knowledge and hands-on clinical skills at a conference such as GLATA. We hope to bring what we have learned back to our university to use on our own LSSU athletes and to our patients that we work with in a variety of settings across this community. Thank you again for not only supporting the profession of athletic training, but for supporting our transition from students to future certified athletic trainers.
EGFR Enzyme System
Chris Savich and Mark Santoyo
LSSU Chemistry and Biology Students
Chris Savich and Mark Santoyo, were awarded a grant from the Fund for LSSU to purchase an EGFR enzyme system, ADP-Glo Kinase Assay Kit, and Staurosporine, so that they could conduct an interdisciplinary research project. The project consisted of Chris Savich synthesizing eight different 3,4-Dihydroquinazolines compounds through novel organic synthetic techniques, and then providing the compounds to Mark Santoyo to test for anti-cancer properties through the inhibition of the EGFR pathway.
Figure 1. Above is a picture of Chris Savich and Mark Santoyo
During the spring and early summer of 2017, Christopher Savich under the advisorship of Dr. R. Adam Mosey employed novel organic synthetic techniques to make a total of eight 3,4-Dihydroquinazolines. 3,4-Dihydroquinazolines are a class of nitrogen containing aromatic compounds that are of particular interest for their pharmaceutical properties, such as anti-fungal, anti-bacterial, and in the case of this study, their possibility for anti-cancer properties. Eight different 3,4-Dihydroquinazolines were made through a novel four step one-pot synthesis. Each of the eight 3,4-Dihydroquinazolines had different functional groups attached to the left side of the molecule so as to test which of the functional groups would produce the greatest effect.
During the summer of 2017, Mark Santoyo under the advisorship of Dr. Martha Hutchens began the second half of the project. Mark’s objective was to test whether any of the eight 3,4-Dihydroquinazoline compounds that Chris synthesized could inhibit the activity of an enzyme called EGFR (epidermal growth factor receptor) kinase. Excessive or uncontrolled activation of this enzyme contributes to the development of certain cancers. EGFR converts ATP to ADP, so if the enzyme was inhibited, less ADP would be produced. First Mark used the ADP-Glo Assay Kit to construct a standard curve relating luminescence intensity to ADP concentration. Next, he used the ADP-Glo kit together with the EGFR Enzyme System to optimize the concentration of EGFR kinase and its substrate for maximum ADP production. Mark then conducted the reaction but introduced a known kinase inhibitor Staurosporine, which was used as a positive control, providing data on how much inhibition would be needed from the 3,4-Dihydroquinazoline compounds to be considered significant. (Figure 2)
Figure 2. Above is a picture of Mark micropipetting a master mix into a 96 well plate.
Once the reference data was established and compounds were ready and purified (Figure 3), the reactions were repeated but instead of using Staurosporine, the 3,4-Dihydroquinazoline compounds were used. ADP production was measured by change in luminescence, read via a microplate reader. The result of this experiment were that the compounds exhibited no significant inhibition of ADP production. However, provided key information that can aid further research in the search for anti-cancer compounds by eliminating compounds with functional groups of the variety tested from the pool of possible functional groups.
Figure 3. Above is a picture of the compounds 51,53, 57 after purification.
In conclusion, Chris and Mark were given the ability to, respectively, utilize and practice organic synthetic chemistry in an applied setting and molecular biology techniques in a manner that came together to further our interdisciplinary understanding of signal transduction pathways, and their importance in current cancer research. We would like to thank the Fund of LSSU Grant for this great opportunity given to us, allowing us to further not only our education but also the understanding of cancer and the possible treatment of it.
Lake-Level Influences on Shoreline Behavior
Christina Boyce and Morgan VanWinkle
LSSU Geology Students
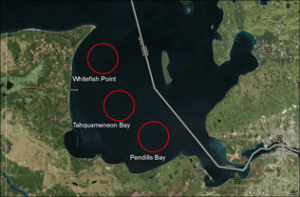
The subsurface architecture of the Grand Marais headland beach along southern Lake Superior, MI, was studied using ground penetrating radar (GPR) and geographic information system (GIS) technologies to determine how lake-level changes have influenced shoreline behavior. Jetties constructed in 1883 to protect the harbor inlet have served as a blockade for sediments transported eastward along the shore by prevailing longshore currents. The resulting buildup of sediment along the west side of the jetty has resulted in near-continuous beach growth at an average rate of 234,136 m³ per year. Aerial photographic reconstructions show this sediment build-up to fluctuate over the decades. Geophysical data was investigated to examine subsurface features and determine whether sediment architectures are influenced by lake level changes.
 The 250 MHz GPR mapped lakeward-sloping sediment units, interpreted as former foreshore deposits, topped by horizontal units, the product of wind processes. Data reveal changes to this basic architecture due to onlap, truncation, and erosion of surfaces. This is caused by influences of different water levels on sedimentation throughout the decades. A beach chronology based on aerial photos, derived from GIS technologies, show that patterns of onlap mapped in GPR coincide with lake-level rise.
The 250 MHz GPR mapped lakeward-sloping sediment units, interpreted as former foreshore deposits, topped by horizontal units, the product of wind processes. Data reveal changes to this basic architecture due to onlap, truncation, and erosion of surfaces. This is caused by influences of different water levels on sedimentation throughout the decades. A beach chronology based on aerial photos, derived from GIS technologies, show that patterns of onlap mapped in GPR coincide with lake-level rise.
Emplacement of the jetty has allowed for preservation of shoreline architectures not accessible elsewhere. Patterns detected from the subsurface data at Grand Marais headland provide useful information on how lake levels influence shoreline dynamics. Additional data collection, particularly seasonal and year-to-year surveys would provide greater knowledge of how short-lived processes factor into shoreline growth.
The final interpretation was presented as a poster at the 2017 Northeastern and North-Central Geological Society of America in Pittsburgh, PA. By gaining experience presenting their data, veteran geologists were able to comment and make suggestions for future work, encouraging the two students to look at the data in a different perspective. Studies at Grand Marais are continuing to gather more information for the area.
Mindfulness and Leadership
Carly Banchiu and Jody Rebek
The LSSU Foundation grant permitted Carly Banchiu, Research Assistant, with the technology required to work with Professor Jody Rebek in conducting a literature review on mindfulness, motivation, leadership, leadership development, and autonomous motivation or self-determination. Both worked together to search and organize literature, and utilized AtlasTi to code and highlight important findings within the peer-reviewed articles they selected. In addtion, each explored mindfulness in their individual contexts. Carly participated in yoga and Professor Rebek practiced daily meditation. Carly and Jody discussed their experiences, along with the outcomes of these mindfulness practices. Collaboratively they discussed the findings and created a synthesis of the findings on each of the topics, along with the implications to learning in higher education. Professor Rebek will be utilizing this information in conjunction with her dissertation research to craft and submit a article for publication in an academic journal.
Superior AquaSystems - Michigan Sustainable Aquaculture
Scott Cooper
With the aid of the Fund for LSSU grant, Superior AquaSystems (SAS) students were able to attend both the North Central Region Aquaculture Center Conference (NCRAC) in Milwaukee, WI and the Seafood Summit in Traverse City, MI.Student comment for NCRAC
Jonathon Edwards: Attending the NCRAC meetings was a priceless learning experience. We got to meet with different aquaculture related companies from all over the United States like Nelson and Pade. The conference was a great way to network with other people in this field from all over the country. There were also many speakers on topics like rearing Arctic Char, various plant beds styles for aquaponics, and much more. We also had a booth for the aquaculture automation challenge youth outreach program. I can’t thank the LSSU Foundation and Fund for LSSU evaluation committee enough for affording our group the opportunity to participate in an event like this.
Scott Cooper: The Wisconsin Aquaculture conference was very beneficial for not only me but the other club members as well. The conference consisted of various talks from Aquafarm owners to researchers that presented data and their findings. In between talks, there was time to engage in conversations with business owners and it was also a great way of networking with possible future employers. We also had the opportunity to have Kacie Ferguson present our Superior Aquasystems Aquaculture Automation challenge along with what SAS is and what we have done with the club over the past year. Funding was greatly appreciated and covered our registration fees for the conference.
High resolution mass spectrometry at Wayne State University
Grace Hubbell
I, Grace Hubbell, along with my advisor Dr. R. Adam Mosey received a grant from the Fund for LSSU to be used to offset the cost of analysis of organic compounds formerly synthesized in our laboratory. These funds provided me with the opportunity to not only receive training on the utilization of a high resolution mass spectrometer, but also allowed me to explore the Wayne State University Chemistry facilities as a possible candidate for graduate school. Because of this financial support by the Fund for LSSU, I received an experience that has enabled me to be more prepared for graduate school and provided a professional development opportunity for me.
On Tuesday, May 3rd, I traveled from Sault Sainte Marie to the campus at Wayne State University. There I was met by Dr. Judy Westrick, a former LSSU professor who is now head of the Lumigen Instrument Center at Wayne State University. Dr. Westrick played an integral part in this opportunity, and not only allowed for analysis and training at a reduced rate, but also graciously offered to host me during my stay.
 After my arrival, I was introduced to the instrument center (left image) and Dr. Yuri Danyliuk, who provided me with training and helped in analyzing my samples. The following three and a half days were spent learning how to analyze the chemical samples in the high resolution mass spectrometer. In my courses at LSSU, I had learned how the practice of mass spectrometric analysis was performed and this experience provided me the opportunity to apply what I had learned in my classes to real life.
After my arrival, I was introduced to the instrument center (left image) and Dr. Yuri Danyliuk, who provided me with training and helped in analyzing my samples. The following three and a half days were spent learning how to analyze the chemical samples in the high resolution mass spectrometer. In my courses at LSSU, I had learned how the practice of mass spectrometric analysis was performed and this experience provided me the opportunity to apply what I had learned in my classes to real life.
Dr. Danyliuk taught me all about the instruments and operating procedure for mass spectrometry chemical analysis. He was also able to show me several other instruments located within the center. By Friday afternoon, I was able to collect and analyze data for around twenty compounds with the help of Dr. Danyliuk. This data was then sent to my advisor via mail so that it could be utilized in research and future publications.
As mentioned before, this experience at Wayne not only taught me how to utilize an important instrument, but it also provided me with the opportunity to discover how larger research institutions function and run. During my stay, I was able to interact with educators and researchers from a wide variety of disciplines and learn more about each. I was also able to view many different areas of the Lumigen Instrument Center and chemistry facilities.
In conclusion, I am very grateful for having had this opportunity that I will carry with me for the rest of my career. This experience would not have been possible without the aid of the Fund for LSSU, and I would like to thank the Fund for its support.
Living at the Edge: Do interstate corridors serve as linear dispersal routes for terricolous lichens?
Stephen Kolomyjec
Thanks to the funding from the LSSU Foundation’s Fund for LSSU grant we were able to purchase Cladonia
rangiferina (grey reindeer lichen) tissue cultures from ATCC.org (the American Type Culture Collection). These initial cultures were used to establish an ongoing axenic (pure) tissue culture of C. rangiferina’s mycobiont. This was a critical step for the eventual goal of sequencing the lichens genome because C. rangiferina, like all lichens, normally exists as a composite organism that combines a fungus (the mycobiont) with an algal and/or bacterial photosynthetic partner (the photobiont – a species of green algae in this case) (figure 1). This symbiosis imparts numerous advantages to the lichens such as incredible hardiness and the ability to colonize habitats long before other form of life.
National Conference for College Women Student Leaders
Melissa Shaffer-O’Connell
“I had a great time at the National Conference for College Women Student Leaders!! The funds I received from LSSU were used towards the conference registration, which I am very grateful for. I attended the conference in June of 2016 and got to attend presentations by famous women, attend workshops on negotiating my salary and recruiting membership in organizations, and to tour Washington D.C. with other young women from Michigan. I hope to go again in 2017 and bring other students with me in the future. Thank you for making this opportunity happen, it was an amazing experience!”
– Melissa Shaffer-O’Connell
Lake State Student Nurses Association - NSNA Conference
Victoria Steffke
The National Student Nurses Association conference allowed the Lake State Student Nurses Association participants to network with prominent individuals, have our resumes looked over, and go to focus sessions on the different types of nursing and the issues going on in the field. The Fund for LSSU grant covered the cost of our registration fees, which enabled us to attend the conference and get an National Council Licensure Examination (NCLEX) review. We feel more confident about taking this licensure exam now. We left feeling inspired about nursing and ready to take action! The information we gained from the conference and also talking with other schools will help us to improve our club as a whole. We are so grateful for the opportunity to attend this conference!
GLATA Annual Meeting
Student Athletic Training Association
 The faculty and students of the Student Athletic Training Association (SATA) would like to thank the LSSU Foundation for the Fund for LSSU grant supporting registration costs for the 2016 Great Lakes Athletic Training Association (GLATA) District 4 Annual Meeting. This year’s conference was held in Wheeling Il, March 9th – 12th offering students and Certified Athletic Trainers the opportunity to learn from Athletic Trainers and other health care professionals from across 6 states of District 4.
The faculty and students of the Student Athletic Training Association (SATA) would like to thank the LSSU Foundation for the Fund for LSSU grant supporting registration costs for the 2016 Great Lakes Athletic Training Association (GLATA) District 4 Annual Meeting. This year’s conference was held in Wheeling Il, March 9th – 12th offering students and Certified Athletic Trainers the opportunity to learn from Athletic Trainers and other health care professionals from across 6 states of District 4.
There were many sessions during the conference offering opportunities to learn and experience assessment and treatment techniques for the shoulder, hip, and ankle including vocal dysfunctions, EEG Imaging, pathophysiological diagnosis, psychological trauma, and much more. Some of the newest equipment for diagnosis of concussions was on display to track neural pathways of the brain, SLOSH theory, and a cervical collar minimizing air pockets in the skull to reducing the movement of brain.
The ability to network with other Athletic Training professionals and enhance our knowledge of the growing field of Athletic Training is vital to student development, something LSSU and the LSSU Foundation take very seriously. Lucas Meehan, a senior in the Athletic Training program said, “I was able to meet athletic trainers who worked with the Chicago Cubs and White Sox, as well as students from across Michigan, Wisconsin and Minnesota. Gaining knowledge and connections through these interactions was a vital experience as we as students begin to get our foot in the door in the professional realm.”
As students representing the University in the public eye, working with International Orthopedics & Sports Medicine, War Memorial Rehab Center, LSSU Health Care Center, and with our own Laker athletes, SATA would like to say thank you for supporting our education as we develop a knowledge base and skills that will propel us successfully into the work force as Athletic Trainers.
 Above is a picture of LSSU Athletic Training Students at the Chicago Bulls NBA
Above is a picture of LSSU Athletic Training Students at the Chicago Bulls NBA
Eastern Fish Health Workshop
Lucas Bradburn
In the words of student, Lucas Bradburn:
“The Eastern Fish Health Workshop really opened my eyes to the job opportunities in the new field of fish health. I met many people in the fish health field as well as those in the business of educating students. It helped to get the perspective of other students especially grad and doctoral students. I also got a good idea of the newest and most upcoming research in the field. I made some good contacts for graduate school and future employers.
Overall this trip helped me advertise LSSU as one of the only universities to have an undergraduate Fish Health program and helped me network with some potential employers.”
Automate 2015
Trace Hill (and fellow LSSU Engineering Students)

LSSU Students selected to attend Automate 2015 pictured in front of LSSU’s conference booth. From left to right: Trace Hill, Eugene Peyerk, Blake Dansfield, Tyler Fontana, and Kerry Pierce.
Five LSSU Engineering students were selected competitively based on academic success and approval from LSSU engineering professors to attend the Automate 2015 conference and trade show. Automate was held in downtown Chicago, IL from March 23-26, 2015 with LSSU students attending on March 25th and 26th.
Attendance of the trade show was free, however the conference sessions required passes which were purchased using the money provided by the Fund for LSSU. The conference sessions provided a refreshing real-world angle on concepts we had studied in our coursework at LSSU. For example, I (Trace Hill) attended a course that discussed the application of nonvisible imaging techniques (such as X-Ray, Ultraviolet, Infrared, etc.) that relied upon many of the theoretical concepts I was learning in my Electromagnetics course. Eugene Peyerk attended a course in image processing that complemented many of the concepts he was studying in his Digital Signals Processing and Digital Design courses.
In addition to providing an enlightening supplement to coursework, the conference sessions gave us a chance to meet and interact with industry professionals. Blake Dansfield had a lengthy conversation with the chief engineer at Clearpath Robotics after attending his presentation on “Advances in Mobile Robotics”. It was opportunities like these that made attending the conference sessions worthwhile for more than just professional training.
When not attending conference sessions, members of our group took turns representing LSSU at our conference booth and exploring the trade show floor, which was full of engaging demonstrations of the newest technology in the industry. One of our favorite demonstrations was Barret Technology’s WAM Arms, which basically allowed a user of one arm to “feel” what the other arm was feeling.
We were the only undergraduate students attending the conference on behalf of their university during the two days we attended Automate. This drew special attention from many of the individuals we interacted with and usually led to some great conversations that showed well on behalf of both ourselves and LSSU altogether. Overall, Automate 2015 was an unforgettable experience that allowed us to explore the latest trends and technology in the industry.
In close, we are very appreciative of getting the opportunity to represent LSSU at the Automate 2015 conference and are very thankful to the Fund for LSSU for supporting this trip.
Electrophoresis Suite
Chemistry Program
With the help of a Fund for LSSU grant, materials were purchased that have and will continue to be used for CHEM 351 (Introductory Biochemistry), CHE M 445
M 445
(Forensic Chemistry), and CHEM 452 (Advanced Biochemical and Molecular Techniques). The purchased electrophoresis equipment has been used extensively for undergraduate research already. Additionally, the purchased equipment found use in the Superior Edventure Camps this summer 2015.
Aside from course work and research, the purchased materials can be utilized for outreach efforts in middle and high school in the future at low cost to LSSU as expendable costs are minimal. Outreach has been planned weekly for the upcoming academic year. The Who Dunnit? program (utilizing materials purchased through this Fund for LSSU Grant and a Meemic Grant), when used during the Superior Edventure Camps, was very well received per camp evaluations and informal comments. A number of high school students enrolled in the camp are considering attending LSSU as a result of their experience. Additionally, the program has allowed LSSU undergraduate students to gain experience performing outreach and volunteering, as well as arranging and conducting laboratory exercises.
The outreach with the students as they have been able to utilize the equipment has been rewarding and memorable.

G.A.M.E. Forum
LSSU Investment Club

Advisor Professor Root along with two students and Dr. Charles Appeadu of the CFA Institute after he presented LSSU with 1st place in the growth division of the student portfolio competition.
The LSSU Investment Club has the opportunity to submit their financial portfolio in a competition against the other universities in attendance at the G.A.M.E. Forum. This year, the LSSU group received first place honors. Congratulations to the students involved in managing that portfolio!
Attending the Global Asset Management Education (G.A.M.E.) forum in New York was an incredible learning experience. The information presented, the lectures and keynote speakers, the networking opportunities and the opportunity to see what it is like to work in the financial industry were some of the many benefits of participating in the forum.
The five members of the Investment Club who attended the event learned from some of the most prominent, high-ranking executives on Wall Street and the financial industry. It wasn’t uncommon to turn on CNBC in our hotel rooms and seethe same people we listened to earlier in the day discussing their investment strategies and market views. It was almost overwhelming to be exposed to people who possess that kind of knowledge and expertise, not to mention having the opportunity to meet them.
The forum also included small breakout sessions which allowed us to explore our individual interests. In these sessions, we not only learned more about the specialized sectors in the financial industry, but also received very useful advice regarding our career path and how to achieve our goals.
The G.A.M.E. forum involves students from 23 countries and more than 100 universities which exposed the members of the LSSU Investment Club to many different points of view. The opportunities we had to meet and establish contacts with people in our field of study will definitely give us an advantage when the time comes to send resumes and get interviews.
Participating in the G.A.M.E. Forum was an eye-opening experience which let us take a look at our professional future and the real life applications of all the theory we learned in the classroom. It also provided us with the incredible advantage of networking and getting advice from very successful people in our desired field. The LSSU Investment Club is very grateful to be given the opportunity to attend the forum and it is our hope that more students get to share this experience in future years.
Community Baby Shower
NURS 325 Course
 The Department of Nursing co-sponsored the annual Community Baby Shower that has been traditionally sponsored only by the War Memorial Hospital OB department. The goal of the Community Baby Shower is to increase education of expectant mothers in our community and the LSSU students taking NUR325 (Nursing Care of the Childbearing Family) were able to fill that educational role.
The Department of Nursing co-sponsored the annual Community Baby Shower that has been traditionally sponsored only by the War Memorial Hospital OB department. The goal of the Community Baby Shower is to increase education of expectant mothers in our community and the LSSU students taking NUR325 (Nursing Care of the Childbearing Family) were able to fill that educational role.
Nursing students in small groups presented evidenced based maternity care practices in 11 different table top displays. Among the topics were breastfeeding, involving fathers, positions for labor, and postpartum depression. The students engaged over 100 community women who were either pregnant or who had recently had a child.
The Fund for LSSU grant allowed for recognition of the role of the Student Nurse and LSSU Nursing in the community. Buttons were purchased that were worn by the WMH OB Registered Nurses that said “I’m a Lake State Nursing Grad”. About 75% of the nursing staff at WMH are LSSU grads! We plan to use those buttons again next year. We also purchased reusable “shopping” totes that had LSSU Nursing on them. The community participants utilized these to collect and bring home all of the information from each of the booths. The Nursing department also shared food costs of the event, as there was a cake and snack table.
The feedback from the NUR325 students was great; they loved the interaction with community members and many were amazed at how many women stopped to talk with them and ask them questions. The students were truly engaged with the participants and were able to put what they had learned in the classroom to practice.
AMA International Collegiate Conference
Shellie Masters
 With help from The Fund for LSSU, Shellie Masters attended the American Marketing Association International Collegiate Conference in New Orleans (March 19-23, 2015) with students, Susan Kirkman and Rachel Rowswell. The students participated in the Marketing Strategy Competition where they placed 4th out of 98 teams. The students had taken 1st Place in an AMA Collegiate regional level competition the previous fall, which was held at Ferris State University.
With help from The Fund for LSSU, Shellie Masters attended the American Marketing Association International Collegiate Conference in New Orleans (March 19-23, 2015) with students, Susan Kirkman and Rachel Rowswell. The students participated in the Marketing Strategy Competition where they placed 4th out of 98 teams. The students had taken 1st Place in an AMA Collegiate regional level competition the previous fall, which was held at Ferris State University.
Susan Kirkman described the competition: “In the Marketing Strategy Competition you are given a case that you must analyze, and come up with a complete marketing plan in just 20 minutes. You are then given 7 minutes to present your plan followed by 2 questions, which you are given 3 minutes to answer.” Susan also commented, “Lake Superior State University was the smallest school in attendance at the conference. This goes to show the small are indeed mighty!”
“The AMA International Collegiate Conference was a wonderful opportunity for our students to practice the skills they have learned at LSSU in a highly competitive environment, as well as participate in a variety of workshops and certification programs to enhance their skills. The energy level throughout the weekend, with over 1,400 students attending the event, was simply amazing! We could not be more proud of Susan and Rachel! ” commented their instructor, Shellie Masters.
The fund assisted in covering conference and travel fees for Shellie Masters.
Study Abroad in Belize
Study Abroad in Belize
Over winter break (December 2015), 15 students enrolled in Dr. Ashley Moerke’s Marine Biology course traveled to Belize (Central America) for 10 days as part of the course lab. Students spent their days immersed in the ecology and culture of coastal and inland Belize, where they snorkeled coral reefs and mangrove ecosystems in the Belize Cayes, and explored fresh waters, cave systems, and rainforests inland. Students also conducting research projects on the diversity of coral reef fishes and invertebrates in Marine Protected Reserves, important conservation areas. In addition to their unique ecological experiences, the group toured two important Mayan archeological sites in western Belize – Xunantunich (pictured) and Cahal Pech. Archaeologists believe that over 200,000 people lived at Xunantunich during its peak around 600 AD. The Fund for LSSU was integral in supporting the cultural visit to Xunantunich, one of the highlights of the trip.
Piping Plover Research
 LSSU Senior Projects
LSSU Senior Projects
The grant from the Fund for LSSU allowed three students, Gislaine Peters, Michelle Kane and Jacob Northuis, to complete their senior thesis Piping Plover research. Piping Plovers are an endangered shorebird that nest on the beaches of the Great Lakes. Game cameras were purchased through this grant and were placed on four beaches in Michigan’s Upper Peninsula: Grand Marais, Gulliver, Port Inland, and Vermillion.
Gislaine Peters
My research centered on the use of wooden huts (cabanas) by piping plover chicks. The plover chicks face difficulties while trying to survive to adulthood, and the cabanas could serve as a safe haven for chicks when they grow too large to hide under their parents. Cabanas were placed at four beaches in the Upper Peninsula. Although we didn’t see a large use of the cabanas in general, we did have one site that used them greatly. Continued use of the cabanas could allow the plovers to become familiar with them and perhaps grant the chicks a greater chance at survival.
Michelle Kane
Piping plover males and females take turns sitting on the eggs, and this project determined who spent more time incubating. Nest monitors recorded number of eggs hatched, chick mortality, chicks fledged and departure date of adults. This information was used to determine how the contributions of each parent during incubation influence overall hatching and fledging success of chicks. Preliminary results indicate that males spend slightly more time on nests and number of chicks fledged is dependent on male incubation.
Jacob Northuis
The data collected by the cameras placed on the nest also allowed for observation of the parental disturbance during incubation. For my senior thesis I am observing the success of the chicks surviving to fledging based on parental disturbance during incubation. I would like to determine if the parents that are more cautious during incubation are more successful when raising their young.
MI Undergraduate Psychology Research Conference
LSSU Psychology Department
 On April 11th, 2015, the LSSU Department of Psychology Senior Research class, 19 students, attended the 28th Annual Michigan Undergraduate Psychology Research Conference hosted by Albion College in Albion, Michigan. This conference invites undergraduate programs in Psychology from Michigan and nearby states to attend. Presenting at this conference is an important experiential learning opportunity for our students and for the past 5 years, it has been a high priority for our program to have our seniors participate.
On April 11th, 2015, the LSSU Department of Psychology Senior Research class, 19 students, attended the 28th Annual Michigan Undergraduate Psychology Research Conference hosted by Albion College in Albion, Michigan. This conference invites undergraduate programs in Psychology from Michigan and nearby states to attend. Presenting at this conference is an important experiential learning opportunity for our students and for the past 5 years, it has been a high priority for our program to have our seniors participate.
We traveled down-state on April 10th in 3 LSSU mini-vans which were covered by the Fund for LSSU grant we received. The conference was held the following day. Of the 19 students that attended, 5 students gave talks while the rest presented posters during the poster session. Our students also had the opportunity to hear undergraduates from several universities in the region present talks and posters. The conference concluded with lunch and an interesting talk by the keynote speaker, Jordan Troisi, PhD from the University of the South.
Overall, this experience was a great learning opportunity for LSSU’s Psychology students. Not only were they able to hear from their peers as well as professionals, but they were able to experience first-hand how to present their own research. This experience will set LSSU students apart as they begin to apply for jobs or as they move on to graduate school.
SWE National Conference
The Society of Women Engineers LSSU Chapter
On October 23rd-26th, 2014, The Society of Women Engineers (SWE) LSSU chapter attended the SWE National Conference in Los Angeles, California. The SWE National Conference hosts the largest career fair in the United States. The majors that were represented include electrical engineering, computer engineering and computer science. The objectives of the trip for the four student attending were to find jobs or internships and to network with professionals.
LSSU students arrived in Los Angeles on October 23rd and attended the conference the following day. At the conference, students attended lectures. For example, three students attended a talk led by an Engineering Manager that works for the CIA. Afterwards, at the career fair, students networked with many companies. Among these companies were GE, NASA, Johnson Controls, Chevrolet, Ford, Honeywell, and AT&T. Jennifer Fredericks was offered a summer position at GE Transportation in Erie, Pennsylvania. Samantha Lies was offered an interview with GE for a position after her graduation this spring. Deborah Rempala talked to NASA and AT&T, and Jessica Tyer met with Honeywell.
Overall, this was a very successful trip. Students gained valuable insights into networking, and students were given opportunities for employment.
GLATA Annual Meeting
 Student Athletic Training Association
Student Athletic Training Association
SATA would like to thank the LSSU Foundation for the Grant supporting registration costs for the recent Great Lakes Athletic Training Association (GLATA) District 4 Annual Meeting in Wheeling, Il. March 11-14. Below are comments as to the value LSSU students gained from attending the GLATA Conference.
Student Comments:
Kaitlyn Lundrigan: “I learned that, as a group of professionals, we need to be more demanding of our own recognition and take charge of our identity and make our knowledge and services more well known. I learned that I really love attending conferences and listening to other people who are experts at what they do. The meeting has inspired me to want to present at a professional meeting.”
Taylor Mills: “I thought GLATA was an amazing experience and I can’t wait to go back next year. I enjoyed the session on the female athlete triad. I enjoyed the presenter’s tips on how to approach an athlete who may have the female athlete triad. You have to be confident and not be too embarrassed to talk to an athlete about such a serious topic.”
Adeline Kariainen: “By attending the GLATA conference, I was able to learn more in depth [information] on functional muscle screening and studies. It was so interesting to see the programs designed by athletic trainers for the athletes and to be able to see pictures of the athletes’ success along with statistics. I also learned about assessing various injuries and if various new rehab techniques work or not. It was a very enjoyable and informative conference, and I would definitely go again.”
Michael Katz: “I learned how posture can affect the back and neck muscles. How for every inch the head sits forward, ten pounds of weight are added to the neck and back muscles. Overall GLATA was a great opportunity to expand our knowledge and update our education as students, as well as network within our profession.”
Advancing Aquaculture in Michigan
Superior AquaSystems
With the aid of the Fund for LSSU grant, Superior AquaSystems (SAS) students were able to attend both the Wisconsin Aquaculture Association Meeting (WAA) in Marshfield, WI and the Michigan Aquaculture Association Meeting (MAA) and Michigan Seafood Summit in Lansing, MI.

LSSU alumnus Dr. Ron Kinnunen of MI Sea Grant presenting on the importance of HACCP training at the MI Seafood Summit.
Scott Cooper: On March 5-7th I attended the WAA conference with Eric Johnston and Dr. Evans. This was a great learning experience and networking opportunity for me. Thursday evening, we chatted with LSSU alumnus Greg Fischer, along with a few other aqua-culturists. Friday we listened to numerous presenters and learned information that will help our aquaponics club with problems at the hoop house. There was also a trade show. I met with the owners of Nelson and Pade Inc. who are my dream employers. Since I was able to attend this conference and meet with Rebecca Nelson, there is a high possibility that I will be interning at their company in Montello, WI this summer. Saturday morning we listened to a two hour aquaponic presentation that was extremely helpful for our club.
Scott Cooper: SAS traveled down to East Lansing for the annual MAA conference. From eight in the morning till nine at night we listened to presentations, learned quality and valuable information that will help SAS build and grow, and networked with future employers. We also shared dinner with two aquaponics owners who have invited us to come and visit their facility. With getting the chance to go to these conferences I, along with SAS, am learning substantial amounts of knowledge towards aquaculture and aquaponics.

MAA meeting and a chance to meet our benefactors. The aquaponics equipment we use was generously donated by the Walled Lake Veterinary Hospital. Pictured L-R are Jon Edwards, Stephen Sajewski, Dr. Steven Burns D.V.M, Lucas Bradburn and Scott Cooper.
Jonathan Edwards: At the MAA meeting and seafood summit, I learned many aspects of aquaculture furthering my interest in this field. We learned about the business, health, types of set up and regulation aspects of aquaponics. One thing that really stuck out is how highly the people involved with MAA spoke of LSSU fisheries graduates. They mentioned how we are one of the only schools to have hands on aquaculture experience. I was able to meet Dan Vogler of Harrietta Hills Hatchery as well as many other private, and hobbyist aqua culturists. We learned valuable knowledge pertaining to our careers and had the chance to network. This was an invaluable experience and I cannot thank Dr. Evans and the school enough for making this all possible.
Exposing Ecosystems through Education and Photography
Angela Gordon
 In the words of student Angela Gordon:
In the words of student Angela Gordon:
For my Senior Experimental Learning Project (ELP), I ran a summer nature photography program for kids aged 6-12 though the Chippewa County 4-H. The program took place June and July of 2014. My program was held for groups of students in either Monday/Wednesday or Tuesday/Thursday sessions for two hours each day. In total, 15 children were a part of the program.
The children visited a different nature location in the county each week. These locations included: Sherman Park, Algonquin Nature Trail, Mission Hill, and Point Iroquois Lighthouse.
Each class focused on a different nature topic. The classes featured environmental learning, hiking, photography, and crafts. We covered subjects such as: photography tips; producers, consumers, and decomposers; lichen; mosquitoes; seed dispersal; life stages of trees; water cycle; water pollution; water bugs and water quality; renewable energy; water 
uses; and invasive species.
I created a website using Shutterfly to advertise and display the photos taken during each class. I uploaded the kids’ photos online to allow them and their parents to see the photos. You can view the students’ photos here: https://connectingkidswithnature.shutterfly.com/pictures.
After my last program was completed, I put together an exhibit of the nature photos in the LSSU Cisler Center displaying the top seven photos taken by each student. There were a total of 105 photos in the showcase. I hosted the exhibition on a Friday night and Saturday morning in mid-August. Attendees during the show included students from the program, their parents, professors, and others.
I received a grant from the LSSU Foundation which I used to buy three digital cameras, SD cards, craft supplies, office supplies, and supplies to protect from mosquitoes.
BAS Epsilon Electrochemical Analyzer
Chemistry Department
Thanks in part to financial support from the Lake Superior State University Foundation via the Fund for LSSU, the School of Physical Sciences at LSSU recently added the BAS Epsilon electrochemical analyzer to their suite of state-of-the-art chemical instrumentation.
Student researchers, under the direction of Dr. Christopher Heth, will use this instrument extensively to study new materials, known as conducting polymers, which will be developed and synthesized in the laboratory at LSSU. These polymers are lightweight and flexible like plastic and have unique electronic properties, which makes them useful in solar cells, organic light emitting diodes (OLEDs), corrosion coatings, and in thin semiconductors.
Students will gain valuable experience with the scientific process by being active participants in real scientific research, as well as develop improved problem-solving skills as they work to make new molecules that have never been synthesized before.
The instrument will also be utilized in several classes. All students enrolled in CHEM 332 (Instrumental Analysis) will gain hands-on experience with electrochemical techniques. Other courses will also use the new instrument as an analysis tool, including CHEM 461 (Advanced Inorganic Chemistry). Chemistry and environmental science students and faculty will also use the analyzer to accurately measure metals in water samples and for other projects.
The acquisition of this instrument sets LSSU apart from the majority of Chemistry programs nationwide, where most undergraduate students’ experiences with electrochemistry are limited to basic activities. The state-of-the-art techniques made possible through the BAS electrochemical analyzer allows students to gain experience with much more advanced aspects of electrochemistry, setting them apart from their peers in other programs.
NSNA Annual Conference
Lake State Student Nurses Association
 The funding provided to our group allowed us to afford attendance to the National Student Nurses Association (NSNA) Annual Conference in Nashville, TN. The conference was an awesome opportunity to network with potential future employers and graduate schools, review for the NCLEX (nursing certification exam), and strengthen our passion for nursing.
The funding provided to our group allowed us to afford attendance to the National Student Nurses Association (NSNA) Annual Conference in Nashville, TN. The conference was an awesome opportunity to network with potential future employers and graduate schools, review for the NCLEX (nursing certification exam), and strengthen our passion for nursing.
Our first two days at the conference were spent in an NCLEX “boot camp” where we learned tips and tricks for test success. During a break on the first day, members of our group were able to participate in an American Red Cross (ARC) Disaster Relief certification course. This training allows us to participate in relief efforts by the ARC should a disaster occur in our area.
Another chunk of our time at conference was spent on professional development. An afternoon was set aside for portraits, mock-interviews, and resume reviews all in an effort to help us land an awesome registered nurse (RN) position fresh out of school! On top of that, we heard from guest speakers about the foundational purpose of the RN in the healthcare team, leadership development, and varying RN specialties including forensic, emergency, and plastic surgical nursing. Additionally, we learned from a session called “Pharmacology Made Insanely Easy” during which we used things like songs and mnemonics to memorize the functions of common medications.
The NSNA conference culminated with a 5K “Fun Run” to raise money for nursing scholarships. Overall, the conference was an excellent experience to learn about our chosen career field and our own interests.
Accounting Club IMA Student Conference
Accounting Club IMA Student Conference
 The LSSU Student Chapter of the IMA (Institute of Management Accountants) was able to represent Lake Superior State University with seven members at the 15th Annual Student Leadership Conference. The conference was held in Indianapolis, IN from November 6-8, 2014.
The LSSU Student Chapter of the IMA (Institute of Management Accountants) was able to represent Lake Superior State University with seven members at the 15th Annual Student Leadership Conference. The conference was held in Indianapolis, IN from November 6-8, 2014.
There were many speakers at the conference from CEO’s to CFO’s and Vice Presidents of companies. Students were able to learn about how to network with the right people, the importance business ethics, how to utilize LinkedIn, and other hot topics in the accounting field. Perhaps the most enjoyed and helpful session the students attended was one that covered resumes. The presenter, Lance Farmer, HRAM, spoke to students about how to format their resume and then tailor the resume for each job application.
Justin Pollard, Accounting Club president, said, “Our whole group benefited from this conference and was able to bring back things we learned to share with other Accounting majors at LSSU. It was very eventful and enabled us to connect to professionals and other students in our field of study.”
Aside from engaging in educational sessions, the students were treated to a trip to the Dallara Indy Car Factory for a tour and to experience race simulations.
“Lake Superior State University was well represented at the 15th Annual IMA Student Leadership Conference. I know that these students will go on to wonderful careers and continue to represent Lake Superior State University in a positive way. Opportunities such as this conference help shape the future for these individuals,” said Accounting Club adviser Jodi Hunter, CMA, CFM.
Funds from their Fund for LSSU award were used to cover registration costs to the conference.
2014 G.A.M.E. Forum
LSSU Investment Club
 Since the LSSU Investment Club’s founding in 1999, students have gathered to enhance their knowledge of stock analysis, economic trends, global financial behavior, and stock portfolio management. The focus and mission of the club is not only to learn more about stock investing, but to create and manage a portfolio that is competitive to the S&P 500 stock index. The club portfolio earned a return of 28.03 % in 2013 and boasted a five year return of 104.78%.
Since the LSSU Investment Club’s founding in 1999, students have gathered to enhance their knowledge of stock analysis, economic trends, global financial behavior, and stock portfolio management. The focus and mission of the club is not only to learn more about stock investing, but to create and manage a portfolio that is competitive to the S&P 500 stock index. The club portfolio earned a return of 28.03 % in 2013 and boasted a five year return of 104.78%.
Ten Investment Club students attended the 2014 Quinnipiac Global Asset Management Education (G.A.M.E.) Forum in New York City from March 20-22, 2014. The three day conference focused on increasing investment knowledge and exposure for club members. It featured many notable representatives from Goldman Sachs, NASDAQ, Bloomberg, CNBC, CFA Institute and many more. The students attended over 15 hours of instruction through keynote panels, workshops and networking opportunities. The conference also provided the opportunity to submit the club’s portfolio in a competition among the other attendees and send one student to represent LSSU during the NASDAQ Closing Bell ceremony on March 19 televised live from Time Square.
Funds received from the Fund for LSSU reduced the cost of conference registration fees, hotel and airfare. The Investment Club is thankful for the support from donors like you who helped make this trip and invaluable educational experience possible.
ASM-MI Conference
LSSU Microbiology Club

Pictured are Microbiology Club students and their advisor Dr. Martha Hutchens. They are standing in front of club member Erin Mulroney’s award winning poster.
On March 28, 2014, students from the LSSU Microbiology Club went to the American Society of Microbiology, Michigan Branch (ASM-MI) conference in Grand Rapids. During the first night, the ASM held a student and faculty mixer which allowed students to meet with professionals in the field of microbiology and talk to other professors and students who shared appreciation of the micro-world.
The theme of the conference was “The Human Microbiome.” This term refers to all of the bacteria, fungi, and viruses that live on or in a healthy human body. The importance of these tiny creatures is only now being properly appreciated; it was amazing to learn about all of the new research going on in the country. Some of the speakers heard included Dr. Joseph Petrosino from Baylor College of Medicine, Dr. Casey Theriot of the University of Michigan, and Dr. Marius Vital from Michigan State University.
Club member Erin Mulroney presented her poster titled “Effects of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonic Acid (PFOS) on Rhodobacter sphaeroides Enzyme Activity.” This poster, co-authored by Dr. Britton Ranson-Olson, won the Best Student Poster Award, bringing recognition to both Erin and LSSU.
The Microbiology Club is grateful to the LSSU Foundation and the Fund for LSSU for supporting this trip. The money received from Fund for LSSU was used for registration fees and to defray the cost of gas for the drive to Grand Rapids.
Equipment Inventory Development
Regional Outdoor Center
 From the ROC’s director Paul Tumey:
From the ROC’s director Paul Tumey:
I would like to thank Lake Superior State University’s Foundation for selecting the Regional Outdoor Center to be the recipient of the $2,500 grant for equipment. The funds were used to purchase cross country ski packages for the upcoming winter. We acquired seven pairs of cross country skis, ten sets of poles, and twelve pairs of various sized boots.
The skis are a great addition to our gear inventory. We will use them for afternoon ski socials around the perimeter of campus, for off campus trips to various locations, and for students to rent. Having the ski packages to rent and offering trips will give students the opportunity to pursue a new outdoor activity or to continue an already existing hobby. I believe the previously mentioned activities, along with others that the Regional Outdoor Center offer, will be attractive to current and prospective students as well as staff and the Sault Ste. Marie community.
Class Excursion
Recreation Studies 365
 Following a semester of learning about risk management, safety, search and rescue, logistics, nutrition, trail techniques, and ethics within the classroom, Dr. Sally Childs’ Recreation Studies 365 class went on an expedition to put what the students learned to the ultimate, real life test. From May 5 to May 14, 2014, eight students and one advisor traveled to Saranac Lake, New York. The goal of the field experience was to gain hands on educational experience and knowledge of hiking, primitive camping, and rafting while getting the chance to enjoy the great outdoors.
Following a semester of learning about risk management, safety, search and rescue, logistics, nutrition, trail techniques, and ethics within the classroom, Dr. Sally Childs’ Recreation Studies 365 class went on an expedition to put what the students learned to the ultimate, real life test. From May 5 to May 14, 2014, eight students and one advisor traveled to Saranac Lake, New York. The goal of the field experience was to gain hands on educational experience and knowledge of hiking, primitive camping, and rafting while getting the chance to enjoy the great outdoors.
This project put everything learned in the classroom into perspective. In the classroom, students learned the importance of preparing oneself for an expedition. They also learned about several theories as they pertain to learning and the ability to perform tasks. One example of a theory taught in the course is the Social Cognitive Theory which states that learning occurs in social context and much of what is learned by a person is learned through observation. The Social Cognitive Theory demonstrates how important an excursion such as this is to allow students to observe how classroom learning can be applied to real life. Overall, the students gained valuable, supplemental education by taking part in this exciting adventure.
Funds from the Fund for LSSU were used to offset the cost of renting canoes for the expedition.
Current status and trends of mercury contamination in Lake Whitefish
William Bernier
The topic that I chose for my senior research project is entitled “Researching the amount of mercury (Hg) in Lake Whitefish Coregonus clupeaformis caught in Whitefish Bay, Lake Superior.” The sampling of the Lake Whitefish took place in the summer of 2013. There were three sampling locations within Whitefish Bay which included Pendills Bay, Tahquamenon Bay and Whitefish Point (Figure 1).
Through laboratory tests and statistical analyses, I discovered the average concentration of Hg in the sampled Lake Whitefish to be 0.0548 ppm (SE + 0.0156). This is a very low quantity and well below the United States Food and Drug Administration’s warning concentration of 1.0 ppm; however, I did find there was a slight increase in concentrations of Hg in Lake Whitefish since 1992 using past data collected from a local agency. The slight increase was not statistically significant, but continued monitoring efforts with larger sample sizes may be able to get a better understanding of why it is slightly increasing.
I chose to work on this project because I am a local resident and I strongly care about our community. Many businesses and families rely on this natural resource. If mercury levels become too high in these fish, the health and security of our community members can become at risk.
I received a grant from the LSSU Foundation to aid in the cost of laboratory expenses, which included extracting the Hg from the fish samples. Thank you for your support.